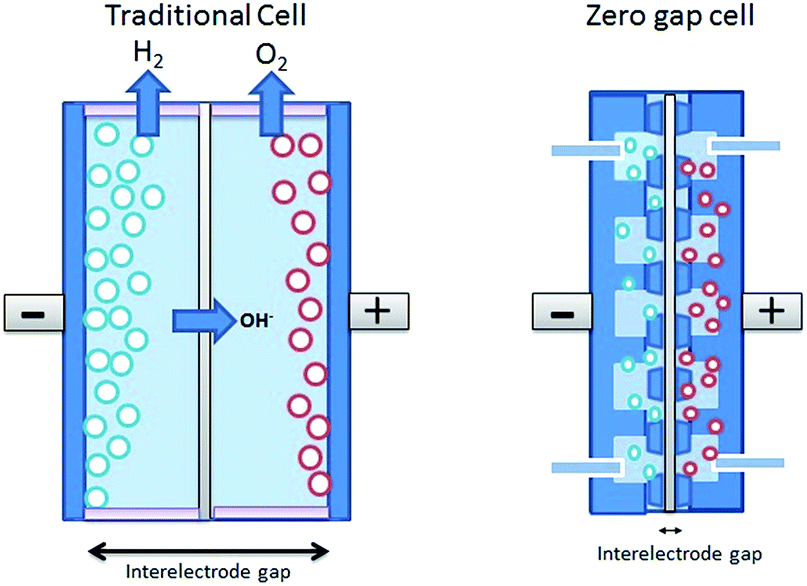
It is possible to use electrochemical processes to increase the alkalinity of seawater, and/or to force the precipitation of solid alkaline materials (i.e., hydroxide minerals). In both approaches, the base and acid streams are recombined and discharged back into the ocean. Basic approaches that force the precipitation of solid carbonate without restoring alkalinity do not result in the net removal of CO 2 from the atmosphere, although they reduce the concentration of CO 2 that is dissolved in solution. Dissolved inorganic carbon is removed and collected as both solid carbonate residues and as evolved CO 2 gas. This creates conditions in which carbonate precipitation can occur and promotes an increase in aqueous CO 2, which may be evolved and collected similarly to the acid approach ( Rau, 2008 de Lannoy et al., 2012). Basic approaches exploit high-pH conditions created around the cathode to shift the equilibrium of the carbonate system toward a greater concentration of bicarbonate and/or carbonate ions ( La Plante et al., 2021). Acid approaches exploit acidic conditions created around the anode to shift the equilibrium of the carbonate system (see Box 8.1) toward a greater concentration of aqueous CO 2, which is evolved/degassed from the solution and collected for permanent storage ( de Lannoy et al., 2012 Datta et al., 2013). Direct CO 2 RemovalĪ range of approaches have been proposed to extract CO 2 from seawater, analogous to methods that remove it directly from the atmosphere ( Willauer et al., 2011 Eisaman, 2020). Several approaches have been proposed that use electricity to promote or to drive reactions that ultimately result in carbon dioxide (CO 2) removal from the atmosphere ( House et al., 2007 Rau, 2008 de Lannoy et al., 2012 Datta et al., 2013 Rau et al., 2013 Eisaman et al., 2018 Zhao et al., 2020 La Plante et al., 2021 Oloye et al., 2021). Technologies use electrochemistry to take measurements of chemical systems, use chemical reactions to generate electricity, or use electricity to drive chemical reactions. Electrochemical Engineering Approaches 8.1 OVERVIEWĮlectrochemistry considers chemical reactions that result in the production or consumption of electricity.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
